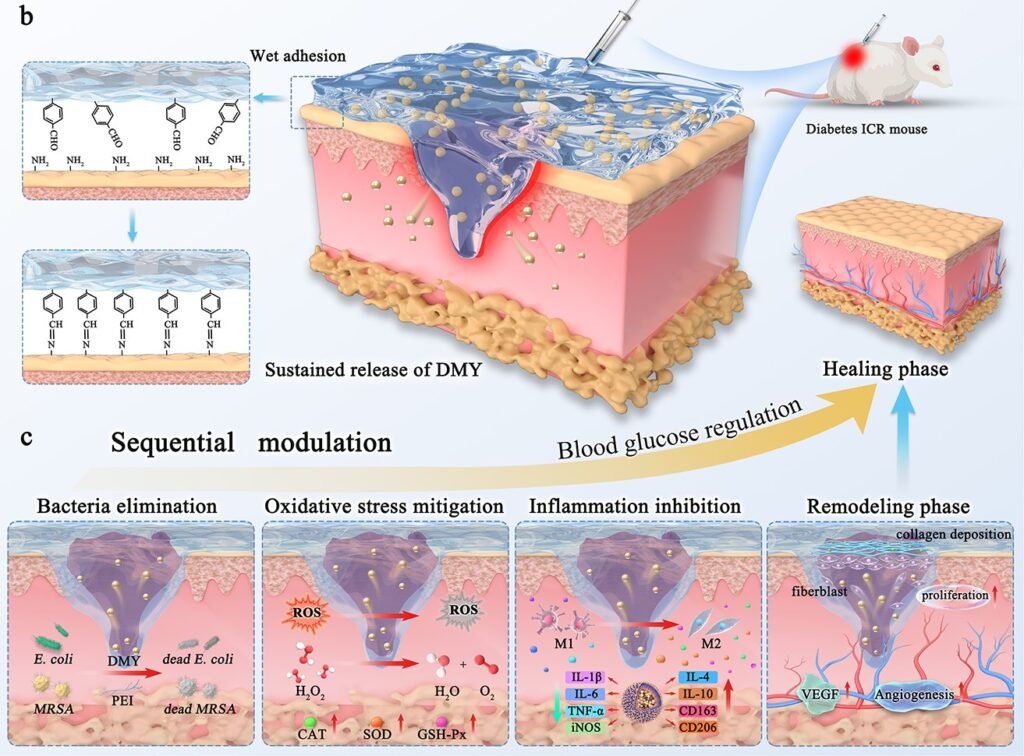
Researchers at Northwest University in China have developed a novel hydrogel treatment that shows significant promise for managing diabetic wounds while also helping regulate blood sugar levels. Chronic wounds are a common and dangerous complication of diabetes, often exacerbated by persistent inflammation, infection, oxidative stress, and poor glycemic control. Traditional treatments typically address these issues separately, but the new hydrogel—referred to as DPFI—offers a multifunctional approach that targets all of them simultaneously.
DPFI is built on a thermosensitive matrix using Pluronic F127, a material that responds to temperature changes to enable controlled release of therapeutic agents. Embedded within this matrix is dihydromyricetin (DMY), a plant-derived compound known for its antioxidant and anti-inflammatory properties. The hydrogel’s design allows DMY to be released gradually at the wound site, delivering a sustained therapeutic effect.
In laboratory and animal studies, DPFI demonstrated a range of beneficial effects. It showed strong antimicrobial activity against common pathogens such as methicillin-resistant Staphylococcus aureus (MRSA) and Escherichia coli, which are frequently implicated in infected diabetic wounds. By reducing bacterial load, the hydrogel helps prevent complications that can delay healing or lead to more severe outcomes.
Beyond its antimicrobial properties, DPFI also reduces oxidative stress—a major barrier to tissue regeneration in diabetic patients. It achieves this by neutralizing reactive oxygen species and promoting a more favorable healing environment. Additionally, the hydrogel modulates the immune response by influencing macrophage polarization. This shift from a pro-inflammatory to a pro-healing state helps resolve chronic inflammation, which is often a persistent issue in diabetic wound beds.
One of the most striking features of DPFI is its ability to promote angiogenesis, or the formation of new blood vessels. This is critical for delivering nutrients and oxygen to damaged tissues, and it plays a key role in accelerating wound closure. In diabetic mice, treatment with DPFI led to significantly faster healing compared to conventional therapies. The wounds showed improved tissue regeneration, reduced inflammation, and enhanced vascularization.
Perhaps most notably, the hydrogel also contributed to better glycemic control in treated animals. This dual action—local wound healing combined with systemic blood sugar regulation—positions DPFI as a potentially transformative therapy for diabetic patients. Rather than treating symptoms in isolation, it offers an integrated solution that addresses both the cause and consequence of impaired healing.