Real‑time blood monitoring has remained out of reach because most sensors quickly clog when exposed to unprocessed blood, losing sensitivity within minutes. This fouling problem has been one of the biggest barriers to continuous biochemical tracking, preventing clinicians from measuring fast molecular changes that could guide personalized treatment. A team led by La Trobe University in Australia has now developed a cell‑inspired sensor that overcomes this limitation by combining a natural protective coating, fast‑responding molecular receptors, and an ultra‑sensitive optical detection method.
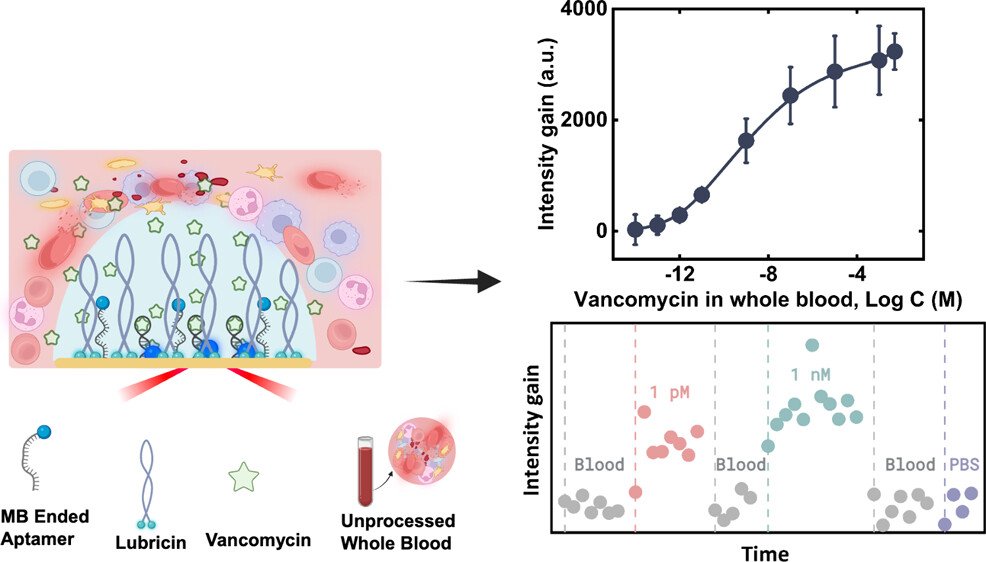
The researchers worked with CSIRO, an Australian Government agency to design a sensor that mimics the way cell surfaces protect themselves while still detecting molecules in their environment. They used lubricin, a naturally occurring protective molecule, to create a microscopic shield that prevents unwanted material from sticking to the sensor. Within this layer, they embedded DNA‑based aptamers that rapidly capture target molecules. This architecture allows the sensor to function inside whole blood without clogging, a challenge that has limited previous technologies.
To read molecular changes, the team used Surface‑Enhanced Raman Scattering, or SERS, an optical technique capable of detecting single molecules. Although SERS is extremely sensitive, it is also highly vulnerable to contamination, which is why it has rarely been used directly in blood. By combining SERS with the lubricin shield and aptamer receptors, the researchers achieved stable, high‑sensitivity detection for more than ten hours of continuous exposure. In a world‑first demonstration, they measured the antibiotic vancomycin in unprocessed blood without any loss of performance.
The researchers explain that the sensor’s cell‑like structure filters molecules from blood while preserving the precision of SERS. Additionally, they note that while other sensors have detected vancomycin, this platform is one hundred million times more sensitive, making it the first practical real‑time SERS sensor capable of operating in a complex fluid like blood. The advance addresses long‑standing challenges in response speed, sensitivity, and surface fouling.
The team emphasizes that the sensor dramatically expands the range of detectable biomarkers, including hormones, toxins, and other low‑concentration molecules important for early disease detection and treatment monitoring. The work also demonstrates a way to overcome the traditional trade‑off between high sensitivity and fast response in molecular testing.
Article from La Trobe University: Cell-inspired sensor paves way to real-time health
Abstract in ACS Sensors: Ultrasensitive, Real-Time Molecular Sensing in Unprocessed Whole Blood Using Surface-Enhanced Raman Scattering Combined with Glycocalyx-Mimicking Structures