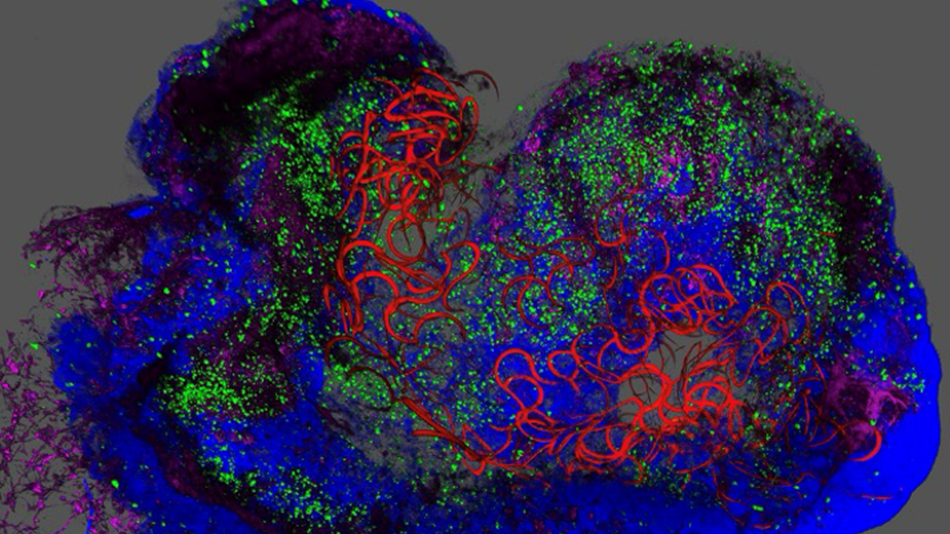
A new study from the University of Pennsylvania reveals that an ultrathin electronic implant can guide lab‑grown pancreatic cells to mature into fully functional insulin‑secreting tissue, offering a potential path toward next generation cell therapies for diabetes. The research aimed to solve a long‑standing problem in regenerative medicine; although scientists can grow pancreatic islet cells from stem cells, these cells often remain immature and fail to release insulin reliably, limiting their therapeutic potential.
The team developed a stretchable, hair‑thin mesh of conductive wires that integrates directly into three dimensional pancreatic organoids as they form. This mesh allows researchers to record electrical activity from individual islet cells and deliver controlled pulses that mimic the body’s natural circadian rhythms. Earlier work from the Alvarez lab showed that immature pancreatic cells respond strongly to rhythmic electrical cues, and the new system allowed the team to apply those cues continuously as the tissue developed. Within days, the cells began cycling on their own, adopting the electrical patterns associated with mature insulin‑producing cells.
The researchers found that the electrical stimulation not only helped individual cells commit to their specialized roles but also encouraged them to synchronize their activity, a hallmark of healthy islet function. This coordinated behavior is essential for proper hormone release, and the study showed that the cyborg‑like tissue secreted insulin at the appropriate times in response to sugar. The mesh also enabled long term monitoring over two months, giving the team unprecedented insight into how human islet cells transition from immature to fully functional states.
The work points to two possible therapeutic directions. One approach would use electrical stimulation during the manufacturing process to prepare lab‑grown islet cells for transplantation, potentially increasing the supply of functional tissue for people with diabetes. Another approach would leave the mesh in place as part of the transplant, allowing ongoing monitoring and stimulation to keep the cells healthy and responsive. The researchers note that future systems could incorporate artificial intelligence to adjust stimulation automatically based on real time cellular behavior.
By combining bioelectronics with stem cell derived organoids, the research teams have created a platform that addresses one of the biggest barriers to lab grown pancreatic tissue. The technology offers a promising foundation for more reliable, scalable, and responsive cell based therapies for diabetes.
Article from UPenn: Could ‘cyborg’ transplants replace pancreatic tissue damaged by diabetes?
Abstract in Science: Implanted flexible electronics reveal principles of human islet cell electrical maturation